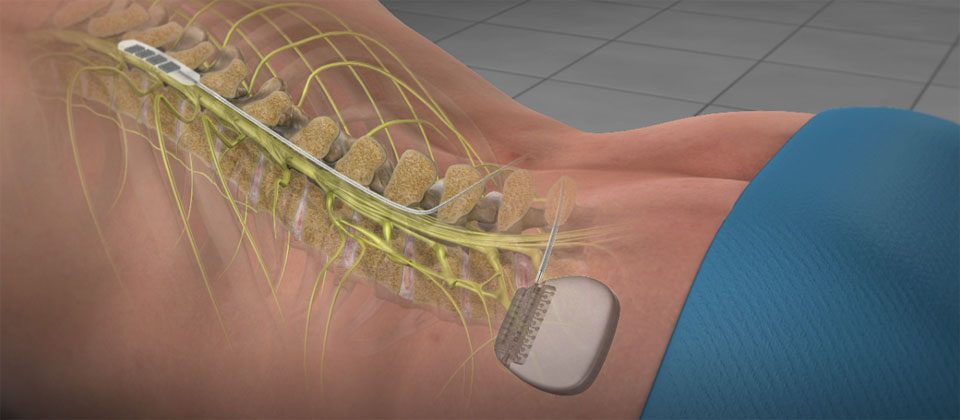
Some patients may be implanted with the components that make up a Magnetic Resonance (MR) Conditional system, which allows them to receive an MRI scan if all the requirements for the implanted components and for scanning are met. Poor surgical risks. Neurostimulation should not be used on patients who are poor surgical risks or patients with multiple illnesses or active general infections. The following warnings apply to this neurostimulation system. Some models of this system are Magnetic Resonance (MR) Conditional, and patients with these devices may be scanned safely with magnetic resonance imaging (MRI) when the conditions for safe scanning are met.įor more information about MR Conditional neurostimulation components and systems, including equipment settings, scanning procedures, and a complete listing of conditionally approved components, refer to the MRI procedures clinician's manual for neurostimulation systems (available online at medical.abbott/manuals).įor more information about MR Conditional products, visit the Abbott Medical product information page at neuromodulation.abbott/us/en/healthcare-professionals/mri-support.html. This system is contraindicated for patients who are unable to operate the system or who have failed to receive effective pain relief during trial stimulation. Indications For UseĪbbott Medical spinal cord stimulation (SCS) systems are indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following: failed back surgery syndrome, nonsurgical back pain (without prior surgery and not a candidate for back surgery), and diabetic peripheral neuropathy of the lower extremities. The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Spinal Column Stimulation (SCS) Systems Intended Use A systematic review and best evidence synthesis of effectiveness of therapeutic facet joint interventions in managing chronic spinal pain. Manchikanti L, Kaye AD, Boswell MV, et al.The impact of chronic pain in the community. Smith BH, Elliott AM, Chambers WA, Smith WC, Hannaford PC, Penny K.Persistent pain and well-being: a World Health Organization study in primary care. Gureje O, Von Korff M, Simon GE, Gater R.Relieving pain in America: a blueprint for transforming prevention, care, education and research. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education.Prevalence of chronic pain and high-impact chronic pain among adults - United States, 2016. Dahlhamer J, Lucas J, Zelaya C, et al.* Patients have reported pain relief following a single RF therapy procedure lasting from 6 to 12 months.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
